Short Communication
Adult Pneumococcal Vaccination: The place of PPSV23 after Launch of PCV20
Varsha Narayanan, Salil Bendre, Prabhakar Dorairaj
Licence:
Attribution-Non-commercial 4.0 International (CC BY-NC 4.0)This license enables reusers to distribute, remix, adapt, and build upon the material in any medium or format for noncommercial purposes only, and only so long as attribution is given to the creator.
Indian Journal of Communicable Diseases 11(1):p 75-80, Jan-Jun 2025. | DOI: 10.21088/ijcd.2395.6631.11225.5
How Cite This Article:
Bendre S, Dorairaj P, Narayanan V. Adult Pneumococcal Vaccination: The place of PPSV23 after launch of PCV20. Indian J Comm Dis. 2025;11(2):75-80.Timeline
Abstract
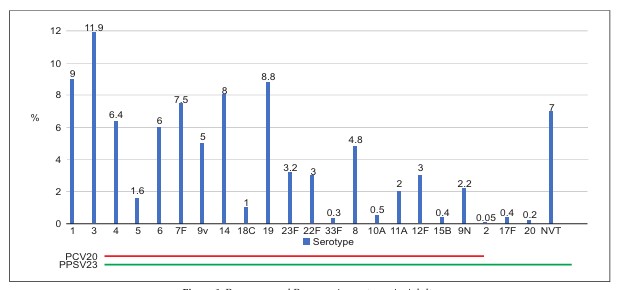
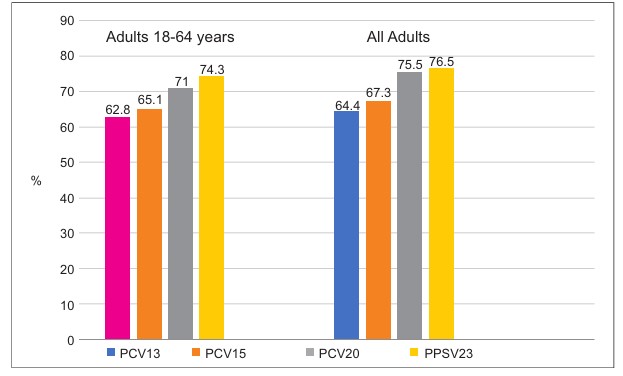
Adult vaccination for pneumococcal pneumonia and invasive pneumococcal disease has faced challenges of low awareness and vaccination rates in India. Both the pneumococcal polysaccharide 23-valent vaccine (PPSV23) and 13-valent conjugate vaccine (PCV13) have been available in India and are part of guidelines and recommendations for elderly adults. Recently PCV20 has also been introduced for adults in India. Given India’s diverse socioeconomic profile, as well as generally lower drive among adults for appreciating the need, and spending on vaccines for themselves as compared to their children, it is imperative to use all available vaccines appropriately in adult pneumococcal vaccination practice. Current and evolving pneumococcal serotype epidemiology in Indian adults, availability of real-world data, cost and access of these vaccines, will determine the future pneumococcal vaccination landscape in our country.
References
- 1. Anderson R, Feldman C. The Global Burden of Community-Acquired Pneumonia in Adults, Encompassing Invasive Pneumococcal Disease and the Prevalence of Its Associated Cardiovascular Events, with a Focus on Pneumolysin and Macrolide Antibiotics in Pathogenesis and Therapy. Int J Mol Sci. 2023 Jul 3;24(13):11038.79
- 2. Pieralli F, Vannucchi V, Nozzoli C, Augello G, Dentali F, De Marzi G, et al; FADOI-ICECAP Study Group. Acute cardiovascular events in patients with community acquired pneumonia: results from the observational prospective FADOI-ICECAP study. BMC Infect Dis. 2021 Jan 25;21(1):116.
- 3. Dhar R, Ghoshal AG, Guleria R, Sharma S, Kulkarni T, Swarnakar R, et al. Clinical practice guidelines 2019: Indian consensusbased recommendations on pneumococcal vaccination for adults. Lung India 2020; 37: S19– S29.
- 4. Koul PA, Swaminathan S, Rajgopal T, Ramasubramanian V, Joseph B, Shanbhag S, et al. Adult immunization in occupational settings: a consensus of Indian experts. Indian J Occup Environ Med 2020; 24: 3-15.
- 5. Koul PA, Vora AC, Jindal SK, Ramasubramanian V, Narayanan V, Tripathi SK et al. Expert panel opinion on adult pneumococcal vaccination in the post-COVID era (NAP-EXPO Recommendations-2024). Lung India 41(4):p 307-317, Jul–Aug 2024.
- 6. Kraicer-Melamed H, O’Donnell S, Quach C The effectiveness of pneumococcal polysaccharide vaccine 23 (PPV23) in the general population of 50 years of age and older: a systematic review and meta-analysis. Vaccine 2016; 34: 1540–1550.
- 7. Tin Tin Htar M, Stuurman AL, Ferreira G, Alicino C, Bollaerts K, Paganino C, et al. Effectiveness of pneumococcal vaccines in preventing pneumonia in adults, a systematic review and meta-analyses of observational studies. PLoS One 2017; 12: e0177985.
- 8. Molander V, Elisson C, Balaji V, Backhaus E, John J, Vargheese R, et al. Invasive pneumococcal infections in Vellore, India: Clinical characteristics and distribution of serotypes. BMC Infect Dis. 2013;13:532.
- 9. Rizvi AA, Singh A. Vaccination coverage among older adults: a population-based study in India. Bull World Health Organ. 2022 Jun 1; 100(6):375-384
- 10. Mrabt, F, Guedes S. Systematic review on serotypes distribution of pneumococcal pneumonia in adults and the elderly. BMC Public Health 25,1194(2025).
- 11. Metcalf BJ, Chochua S, Walker H, Tran T, Li Z, Varghese J, et al. Invasive Pneumococcal Strain Distributions and Isolate Clusters Associated with Persons Experiencing Homelessness During 2018, Clinical Infectious Diseases, June 2021; 72(12): e948–e956.
- 12. Zhao C, Zhang F, Wang Z, Yang S, Chen H, Wang H. Distribution of serotypes and antibiotic resistance profiles of Streptococcus pneumoniae in hospitalized adult patients: a retrospective multicenter surveillance in China (2018–2019). BMC Infect Dis 2025; 25: 980.
- 13. Varghese R, Yesudhason BL, Vimala LR, Neeravi A, Anandhan K, Baskar P, et al. Pneumococcal serotypes causing non-invasive pneumonia in adults from a South Indian tertiary care hospital and the impact of the newer conjugate vaccines. Access Microbiol. 2021 Dec 7;3(12):000258.
- 14. Nagaraj G, Govindan V, Ganaie F, Venkatesha VT, Hawkins PA, Gladstone RA, et al. Streptococcus pneumoniae genomic datasets from an Indian population describing prevaccine evolutionary epidemiology using a whole genome sequencing approach. Microb Genom. 2021 Sep;7(9):000645.
- 15. Kim HW, Lee S, Kim KH. Serotype 6B from a pneumococcal polysaccharide vaccine induces cross-functional antibody responses in adults to serotypes 6A, 6C, and 6D. Medicine (Baltimore). 2016 Sep;95(37):e4854.
- 16. Szu SC, Lee CJ, Parke JC Jr, Schiffman G, Henrichsen J, Austrian R, et al. Crossimmunogenicity of pneumococcal group 9 capsular polysaccharides in adult volunteers. Infect Immun. 1982 Mar;35(3):777-82.
- 17. Feemster K, Hausdorff WP, Banniettis N, Platt H, Velentgas P, Esteves-Jaramillo A, et al. Implications of Cross-Reactivity and Cross-Protection for Pneumococcal Vaccine Development. Vaccines (Basel). 2024 Aug 28;12(9):974.
- 18. Ngamprasertchai T, Ruenroengbun N, Kajeekul R. Immunogenicity and Safety of the HigherValent Pneumococcal Conjugate Vaccine vs the 13-Valent Pneumococcal Conjugate Vaccine in Older Adults: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Open Forum Infect Dis. 2025 Feb 5;12(2):ofaf069.
- 19. Essink B, Sabharwal C, Cannon K, Frenck R, Lal H, Xu X, et al. Pivotal Phase 3 Randomized Clinical Trial of the Safety, Tolerability, and Immunogenicity of 20-Valent Pneumococcal Conjugate Vaccine in Adults Aged ≥18 Years. Clin Infect Dis. 2022 Aug 31;75(3):390398. Erratum in: Clin Infect Dis. 2024 Nov 22;79(5):1327.
- 20. Haranaka M, Young Song J, Huang KC, de Solom R, Yamaji M, McElwee K, et al. A phase 3 randomized trial of the safety and immunogenicity of 20-valent pneumococcal conjugate vaccine in adults ≥ 60 years of age in Japan, South Korea, and Taiwan. Vaccine. 2024 Feb 15;42(5):1071-1077.
- 21. Pérez-García C, González-Díaz A, Domenech M, Llamosí M, Úbeda A, Sanz JC, et al. The rise of serotype 8 is associated with lineages and mutations in the capsular operon with different potential to produce invasive pneumococcal disease. Emerg Microbes Infect. 2025 Dec;14(1):2521845.
- 22. Hurley D, Griffin C, Young M, Scott DA, Pride MW, Scully IL, et al. Safety, Tolerability, and Immunogenicity of a 20-Valent Pneumococcal Conjugate Vaccine (PCV20) in Adults 60 to 64 Years of Age. Clin Infect Dis. 2021 Oct 5;73(7):e1489-e1497.
- 23. Varghese R, Gurumoorthy M, Thomas GG, Walia K, Kumar A, Qureshi S, et al. Accelerating pneumococcal protection in India: The case for rapid adoption of PCV20 across pediatric and adult populations. Indian J Med Microbiol. 2025 Jul-Aug;56:100893.
- 24. CDC [Internet]. Summary of Risk-based Pneumococcal Vaccination Recommendations. May 2025. Accessed Sep 2025. Available on https://www.cdc.gov/pneumococcal/hcp/vaccine-recommendations/risk-indications. html
- 25. Bhardwaj P, Dhar R, Khullar D, Rath P, Subramanian S, Tiwaskar M, et al. Role of Pneumococcal Vaccination as a Preventative Measure for At-risk and High-risk Adults: An Indian Narrative. J Assoc Physicians India 2025;73(5):45-54.
Data Sharing Statement
Funding
Author Contributions
Ethics Declaration
Acknowledgements
About this article
Cite this article
Bendre S, Dorairaj P, Narayanan V. Adult Pneumococcal Vaccination: The place of PPSV23 after launch of PCV20. Indian J Comm Dis. 2025;11(2):75-80.
Licence:
Attribution-Non-commercial 4.0 International (CC BY-NC 4.0)This license enables reusers to distribute, remix, adapt, and build upon the material in any medium or format for noncommercial purposes only, and only so long as attribution is given to the creator.
| Received | Accepted | Published |
|---|---|---|
| October 03, 2025 | October 14, 2025 | December 30, 2025 |
DOI: 10.21088/ijcd.2395.6631.11225.5
Keywords
PneumococcalPPSV23PCV13PCV20SerotypeAdult VaccinationSearch for Similar Articles
Similar Articles
- Emergence of Rabies Cases in Kerala: A Public Health Concern
- Emerging Threat of Microplastic Pollution: A Comprehensive Review
- Climate Change, Extreme Weather Events and Infectious Disease risk: A Narrative...
- Patients Behaviour During an Emergency Situation of a Pandemic and Recommendatio...
- Herpes Zoster Virus Infection of Facial Nerve Treated Successfully without Corti...
Article Level Metrics
Last UpdatedSunday 01 March 2026, 09:10:33 (IST)
Accesses
Citations
Download citation
Article Keywords
Keyword Highlighting
Highlight selected keywords in the article text.
Timeline
| Received | October 03, 2025 |
| Accepted | October 14, 2025 |
| Published | December 30, 2025 |
licence
This license enables reusers to distribute, remix, adapt, and build upon the material in any medium or format for noncommercial purposes only, and only so long as attribution is given to the creator.